To evaluate a mushroom extract powder supplier, supplement brands should start by requesting batch-specific COAs and independent third-party lab reports — then systematically verify the following:
Formulation performance: Test pilot samples for solubility, taste, flowability, and stability under your actual production conditions before committing to volume.
- Species identity: Confirm the exact mushroom species using DNA barcoding; mislabeling is widespread in this category and directly impacts label claims.
- Third-party testing: Review results for heavy metals, pesticides, aflatoxins, microbiology, and residual solvents — not just the supplier’s own COA.
- Quality systems: Verify current GMP certification, audit readiness, and documented change control; these predict batch-to-batch consistency far better than a single sample.
- Extraction & standardization: Confirm the extraction method (hot water, dual extraction), active marker targets such as beta-glucans, and the test method used — “polysaccharides” alone is not a sufficient spec.
- Traceability: Require farm-to-batch lot records and retention samples; without these, recalls become unmanageable.

Mushroom Extract Supplier Checklist: Quality, Safety & Compliance
Use this checklist to validate whether a supplier can consistently deliver safe, standardized mushroom extract powders suitable for dietary supplements:
- Species authentication
- DNA barcoding to confirm the declared species (e.g., Ganoderma lucidum, Cordyceps militaris).
- Part used declared and controlled: fruiting body vs mycelium vs “biomass,” plus substrate/grain disclosure when applicable.
- Active marker and standardization
- Clear targets such as beta-glucans (method specified), triterpenes (for reishi), or cordycepin (for C. militaris).
- Specification includes test method references (e.g., AOAC, USP, validated in-house method) and acceptance limits with realistic tolerances.
- Third-party testing and documentation
- Batch-specific COA plus independent lab report available on request.
- Contaminant panel appropriate to supplements: heavy metals, pesticides, PAHs (if relevant), microbes (incl. Salmonella/E. coli), aflatoxins/ochratoxin, residual solvents (if solvent extraction), and radiological testing where risk warrants.
- GMP and quality system maturity
- Current cGMP certification (dietary supplement/food grade as applicable), supplier audits, CAPA history, change control, deviation handling.
- Identity testing on incoming botanicals/fungi and finished extract release against written specs.
- Regulatory fit and claim support
- Proper product classification (dietary ingredient vs novel food/other jurisdictional requirements).
- Claims are supported by compositional data; avoids unsupported “drug-like” positioning in sales materials.
- Allergen, adulteration, and cross-contamination controls
- Allergen statement, gluten controls (especially where grain substrate is used), and a formal fraud vulnerability/adulteration program.
- For mushroom extracts specifically, the adulteration risk to watch is carrier inflation — maltodextrin added beyond functional levels to reduce per-kilogram cost. Ask for carrier type and percentage to be declared on the COA, and confirm whether a carrier-free option is available if your formulation requires it.
- Traceability and supply continuity
- Lot-level traceability to cultivation site and harvest; retention samples and recall readiness.
- Capacity, lead times, MOQ flexibility, and dual sourcing plans for critical SKUs.
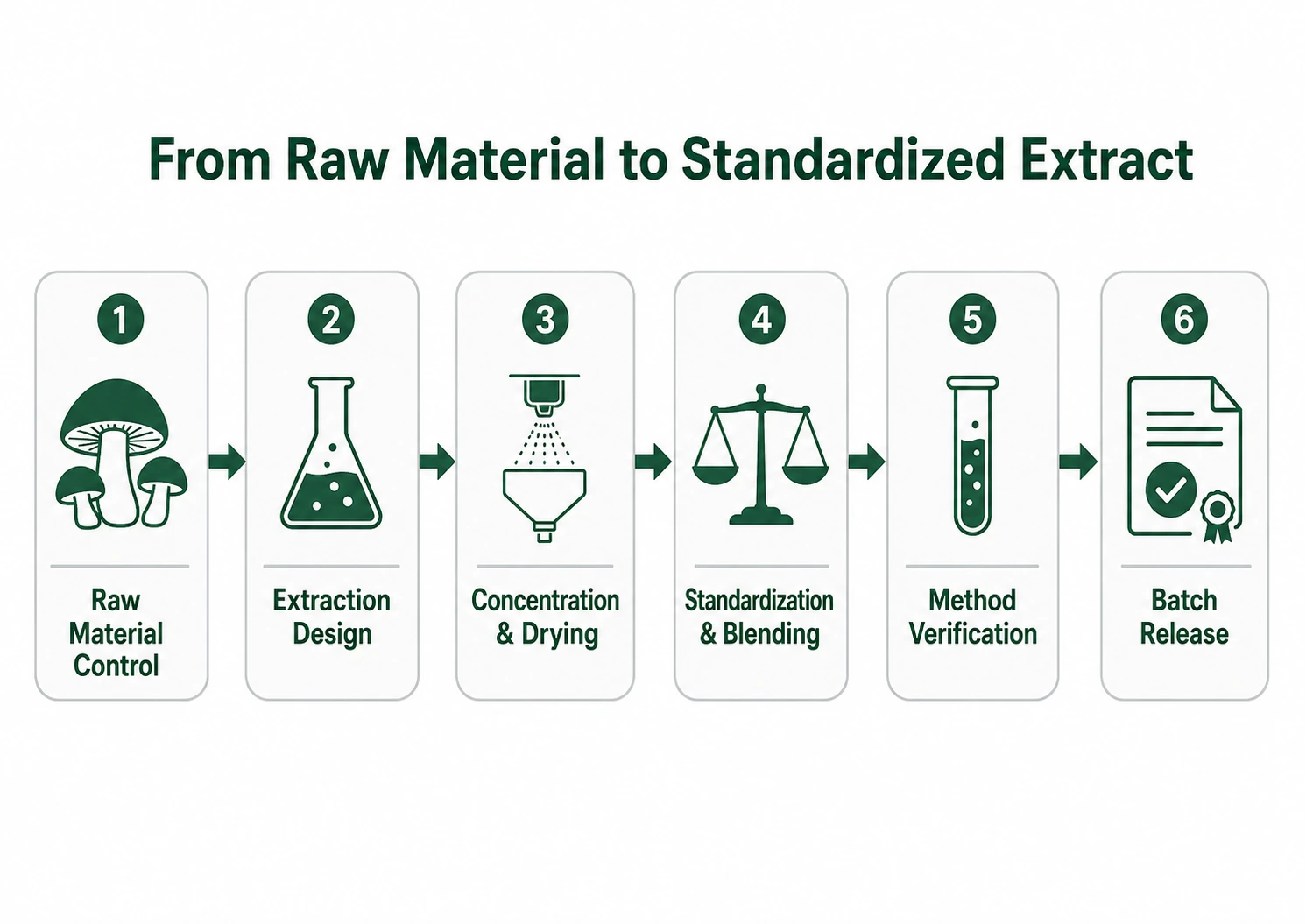
How to Verify Mushroom Extract Quality: Extraction, Standardization & Testing (What “Good” Looks Like)
A credible mushroom extract supplier should be able to explain and document the full technical pathway from raw material to standardized powder. Evaluate it as a process:
- Raw material controls
- Defined strain/species, cultivation method, harvest window, drying parameters, and storage conditions that preserve polysaccharides and minimize microbial load.
- Extraction design
- Hot water extraction commonly targets polysaccharides; dual extraction (water + ethanol) is often used to capture both polysaccharides and alcohol-soluble fractions (e.g., triterpenes).
- Declared solvent system, time/temperature, and solid-to-liquid ratio; residual solvent control if ethanol/other solvents are used.
- Concentration and drying
- Concentration under controlled conditions and drying method (e.g., spray-dried) with moisture and water activity targets to prevent spoilage.
- Standardization and blending
- Blending to meet marker specs should be transparent and backed by in-process testing; avoid “spiking” concerns by ensuring the marker reflects the natural fraction.
- One specific additive worth asking about directly is maltodextrin — a common carrier used in spray-dried extracts. Regulatory guidelines permit a certain percentage in finished extract powders (typically 5–10%), and at these levels it serves a legitimate processing function. However, some manufacturers add maltodextrin at significantly higher concentrations — 40–50% — which dilutes active compound concentration while inflating powder volume. The result is a product that looks like an extract but delivers a fraction of the active marker content per gram.
- There are two ways to verify this: request that the COA specifies the carrier type and percentage, and ask whether the supplier can produce a maltodextrin-free extract using the original mushroom powder as the carrier matrix instead. A manufacturer with genuine extraction capability will say yes. This is a custom option we offer specifically for buyers who want to eliminate any carrier concerns — the mushroom powder itself becomes the base, and the extracted actives are standardized against it. It’s a cleaner label story and removes the blood sugar concerns some consumers associate with maltodextrin.
- Method integrity for beta-glucans
- The supplier should specify whether beta-glucans are tested by enzymatic methods that distinguish alpha- vs beta-glucans; ambiguous “polysaccharides” specs are less informative.
- Batch release and stability
- Defined release criteria and evidence of marker stability under typical storage conditions; stability protocols should match your packaging and climate.
Mushroom Extract Supplier Scorecard: How to Compare & Choose(Commercial + Technical Fit)
Use a weighted scorecard to compare short-listed vendors and avoid choosing on COA and price alone:
| Evaluation Area | What to Verify | Why It Matters | Pass Indicators |
|---|---|---|---|
| Identity & Part Used | DNA ID, fruiting body vs mycelium, substrate disclosure | Prevents mislabeling and potency drift | DNA report + clear BOM and labeling alignment |
| Standardization | Marker targets + validated methods | Ensures repeatable efficacy positioning | Tight specs, method references, sensible tolerances |
| Contaminants | Full panel + third-party lab access | Reduces recall and compliance risk | Low results with jurisdiction-appropriate limits |
| GMP & QA Systems | Certifications, audit openness, change control | Predictable quality and fewer surprises | Audit-friendly, mature CAPA, documented controls |
| Traceability | Farm/producer to batch, retention samples | Faster investigations, reliable continuity | Lot genealogy + retention program + recall SOP |
| Formulation Performance | Solubility, taste, color, flowability, density | Impacts manufacturing and consumer experience | Bench samples + application data + consistent organoleptics |
| Commercial Terms | MOQ, lead time, pricing stability | Prevents stockouts and margin shocks | Predictable lead times + transparent cost drivers |
A Compliance Blind Spot That Pulls Products Off Shelves
One scenario we’ve seen play out more than once: a brand sources mushroom extract powder from China, clears standard COA requirements, ships product to the US — and then gets a compliance notice in California.
California’s Proposition 65 sets lead limits significantly stricter than federal FDA thresholds. For mushroom extract products, the threshold for lead is 0.5 micrograms per daily serving. A product that tests clean under Chinese pharmacopeia standards — which use AAS (atomic absorption spectroscopy) for heavy metal detection — can still trigger Prop 65 violations when tested under ICP-MS, the higher-sensitivity method standard in US and EU labs. The instrument difference alone can expose a gap that wasn’t visible on the original COA.
The brand that sourced elsewhere had to pull inventory. The brands sourcing from us didn’t — because we test to ICP-MS standards before shipment, specifically because we know where our customers sell.
If you’re selling into California, ask your supplier directly: which heavy metal detection method do you use, and have your products been tested against Prop 65 limits? An evasive answer is informative.
A rigorous checklist plus a simple scorecard helps brands select mushroom extract powder suppliers that are reliable, compliant, and formulation-ready.
Molai Biotech Pro Tip: We found the fastest way to uncover weak suppliers is to request three non-consecutive batch COAs plus the underlying third-party contaminant reports, then run a small pilot blend—lot-to-lot changes usually show up immediately in flowability, taste, and beta-glucan variance.

From a Few Kilograms to 2,000kg a Year: A Brand-Building Case
Not every buyer comes to us with a quality problem to solve. Some come with an idea and no supply chain behind it yet.
A few years ago, a buyer from Canada reached out — early stage, exploring whether a functional mushroom brand was viable in their market. Their first order was small. A few kilograms, enough to develop product and test the market response.
We didn’t treat it as a small order. We provided the same documentation — COAs, third-party lab reports, full traceability — that we’d provide to a buyer taking a container. Because the buyers who grow are the ones who started with the right foundation.
Three years later, that same buyer is ordering close to 2,000 kilograms annually. They built a brand, found their customers, and scaled. We grew with them.
The product works when the extract is real. Their customers came back. That’s the only reason the volume grew.
If you’re at the early stage of building something, the supplier decision matters more, not less. You don’t get a second chance to make a first impression on your own customers.

Frequently Asked Questions
What certifications should a mushroom extract powder supplier have?
A qualified supplier should hold at least cGMP certification for dietary supplements or food-grade manufacturing. Additional certifications to look for include USDA Organic, ISO 22000, Kosher, and Halal. For export to the US and EU markets, ask whether the facility has undergone third-party audits and whether they can provide audit reports on request.
How do I verify the beta-glucan content in a mushroom extract?
Request the test method used alongside the beta-glucan figure. The most reliable approach uses an enzymatic method that distinguishes beta-glucans from alpha-glucans — a spec that only states “polysaccharides” is far less meaningful. Ask whether the method is AOAC-referenced or a validated in-house method, and request the underlying third-party lab report, not just the supplier’s own COA.
What is the difference between fruiting body and mycelium extract?
Fruiting body extract is made from the actual mushroom, which naturally contains higher concentrations of active compounds such as beta-glucans and triterpenes. Mycelium extract is often grown on grain substrate, which means the final powder can contain significant amounts of starch with lower active marker levels. Always ask the supplier to declare the part used and disclose the substrate — a reputable supplier will do this transparently.
The regulatory picture adds another layer. In the EU, mycelium-based products are not recognized as equivalent to fruiting body extracts for supplement classification purposes — meaning a product made from mycelium cannot legally be positioned the same way as a fruiting body extract in European markets. In the US, the FDA takes a more permissive stance and recognizes both. China’s regulatory framework similarly permits mycelium-derived pharmaceutical compounds.
The practical implication for buyers: if you’re selling into Europe, confirm explicitly that your supplier is using fruiting body material — and ask for DNA verification to confirm species identity, since Ganoderma lucidum (the only Reishi species approved for use in Chinese health products under NMPA guidelines) looks nearly identical to several related species that don’t carry the same regulatory or efficacy standing.
How many COAs should I request before approving a new supplier?
Request at least three batch-specific COAs from non-consecutive production runs, along with the corresponding independent third-party lab reports for each batch. This reveals lot-to-lot consistency in active markers and contaminant levels — a single COA tells you very little about what you will consistently receive at scale.
One additional step: don’t let the supplier choose which batches to show you. Ask to specify the lot numbers yourself, or ask for the most recent three consecutive batches. Suppliers who only volunteer their best-performing batches are telling you something about their consistency.
What contaminants should be tested in mushroom extract powder?
A complete contaminant panel for supplement-grade mushroom extract should cover heavy metals (lead, arsenic, cadmium, mercury), pesticide residues, aflatoxins and ochratoxin A, microbiological indicators including Salmonella and E. coli, and residual solvents if ethanol extraction is used. Suppliers targeting the US or EU market should test to limits that meet FDA or EFSA requirements respectively.
How do I evaluate a supplier’s traceability system?
Ask the supplier to provide lot-level traceability documentation that traces each batch back to the cultivation site and harvest date. A reliable supplier will also maintain retention samples for each production batch and have a documented recall procedure. If a supplier cannot provide this information readily, it is a significant red flag for quality system maturity.
